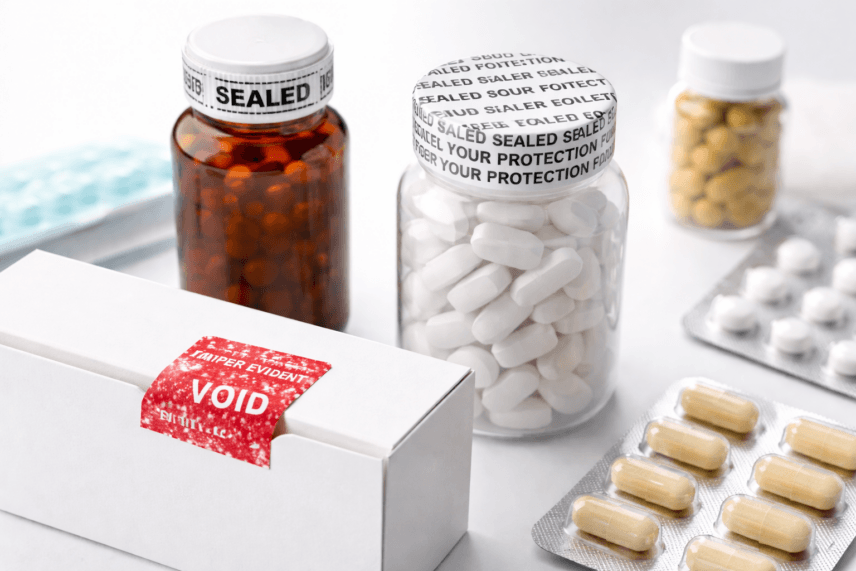
 Tamper-evident packaging (TEP) is widely used across pharmaceutical and supplement product packaging to provide visible evidence if a package has been opened, altered, or compromised, adding a layer of security for both producers and consumers. This is distinct from tamper-proof packaging, which is designed to prevent access rather than simply reveal that access occurred.
Tamper-evident packaging (TEP) is widely used across pharmaceutical and supplement product packaging to provide visible evidence if a package has been opened, altered, or compromised, adding a layer of security for both producers and consumers. This is distinct from tamper-proof packaging, which is designed to prevent access rather than simply reveal that access occurred.
With tamper-evident packaging, manufacturers and their contract packaging partners use features designed to provide clear visual proof that a package has been tampered with or that its integrity has been compromised. A number of different methods get the job done for contract packaging companies working with pharmaceuticals, nutraceuticals, and supplements.
How Tamper-Evident Packaging Protects Products
Protecting package contents from tampering falls on pharmaceutical and supplement producers, relying on experienced turnkey packagers to maintain high standards safeguarding consumers. The responsibility is monitored and mandated by Food and Drug Administration (FDA) regulations, ensuring proper practices.
Packaging requirements for over-the-counter (OTC) drug products intended for human consumption, for example, are regulated by the FDA under the federal Food, Drug, and Cosmetic Act. The Act empowers the FDA to establish packaging standards for covered OTC drug products, helping enhance security and support consumer safety. Covered OTC drug products that fail to meet applicable tamper-evident requirements may be considered adulterated under federal law.
Beyond compliance and liability considerations, tamper-evident packaging also strengthens consumer trust. In order to meet FDA regulatory thresholds, tamper-evident materials cannot be reattached or reapplied without leaving clear evidence the package has been breached. When applying the standard to consumer supplements and pharmaceuticals, contract packaging professionals also account for end-user perceptions and preferences relating to package designs.
Why Demand for Tamper-Evident Packaging Is Growing
The tamper-evident packaging market represented just over 2.5 billion USD in 2025. Forecasts anticipate more than 10% CAGR through 2030, by which time the annual market is expected to exceed 4.25 billion dollars. Regulatory compliance mandates are responsible for some of the projected growth, reflecting changes in drug-traceability requirements. Once voluntary pilot programs have now evolved into binding pharmaceutical packaging standards governing unit-level tracking and tamper-evident features. The full force of the US Drug Supply Chain Security Act calls for visible tamper evidence on each pharmaceutical unit, reflecting an ongoing requirement mirrored by the EU Falsified Medicines Directive, which now applies to 27 overseas markets.
“We take this action with great reluctance and a heavy heart. But since we can’t control random tampering with capsules after they leave our plant, we feel we owe it to consumers to remove capsules from the market.”
—James Burke, Chairman of Johnson & Johnson, at a 1986 press conference on Tylenol capsules
Consumer behavior is also at work driving the increased use of tamper-evident packaging (TEP) for drugs and supplements. Not only do modern consumers demand transparency and high food safety standards across international supply chains, but buyers have also changed the way they purchase products like OTC medication and health supplements. Ecommerce growth, resulting in double-digit parcel increases in 2024, creates safety vulnerabilities as packages are stored, shipped, and ultimately reach consumer mailboxes. In order to reduce unnecessary exposure, Amazon and other major retailers have adopted tamper-evident solutions that illustrate package integrity throughout the delivery process.
Logistics providers and insurers also play a role in the rise of tamper-evident innovation, accounting for packaging security measures in their rates and ratings. And specific tampering incidents, such as an Asian counterfeit baby formula breach, have further cemented tamper-evident packaging as a worldwide standard across health and personal care markets.
Common Types of Tamper-Evident Packaging
Digital security measures are applied to some pharmaceuticals, using blockchain technology to track product integrity, but the practice has limitations, which will likely prevail until the cost and availability of digital solutions renders the strategy more accessible and affordable. In the meantime, tamper-evident packaging commonly relies on tape, seals, labels and special blister packs to enhance the safety and security of package contents.
Shrink bands and shrink seals are among the most widely used tamper-evident solutions for OTC packaging. Torn or broken seals indicate a package has been opened, resulting in visible confirmation from package features that cannot be restored. Obvious visual proof of tampering is also achieved using designs that include plastic pieces that break and alert consumers when product packaging integrity is suspect.
Visual messaging is also deployed, showing statements and icons that only appear after a package seal has been breached. Security labels and tamper-evident tapes offer flexible options for common drug and nutraceutical formats, including pill bottles, blister cards, ecommerce mailers, and secondary packaging.
Key Benefits of Tamper-Evident Packaging
Once considered a premium feature, tamper-evident packaging has become an expected standard for food, drugs, and supplements. Pharmaceuticals and related industries still account for the largest share of the tamper-evident packaging market, but the safety measure is now deployed across widespread consumer categories, from meal delivery to Amazon shipments. Consumers overwhelmingly support the technology and expect to see it used by brands they trust.
Among the primary benefits of tamper-evident packaging, the technology helps manage overall production costs and reinforces brand confidence. Incorporating visible features illustrating package integrity decreases the likelihood and incidence of product contamination, including problems arising from deliberate criminal acts. As far back as the 1982 Tylenol tragedy, when several individuals lost their lives after ingesting poisoned pain relievers, tamper-evident seals have added extra layers of security to protect end users from criminal harm. Serving as a line of defense against similar disasters, TEP enhances public safety while reducing liability costs associated with catastrophic tampering.
In addition to individual unit applications, tamper-evident tape is increasingly used on secondary packaging and shipping cartons. The strategy further protects brands and consumers from counterfeiting, theft, and adulteration, ultimately facilitating consumer confidence and building brand loyalty. Tamper-evident packaging designs also reduce loss and theft expense in retail settings, mitigating the temptation to “try before you buy” or steal individual units from supplement and OTC medicine packages. Compromised TEP offers visual proof that contents may have been exposed to conditions accelerating degradation and reducing their effectiveness.
Experienced contract packagers understand how to apply tamper-evident solutions in ways that support compliance, protect product integrity, and give consumers clear visual confirmation.
